Hot Science Experiments Posts


How To: Suck a Hard-Boiled Egg into a Bottle with a Burning Match
Can you suck an egg into a bottle? Let's find out. For this quick science experiment, you will need an egg, a bottle, and matches. Simply drop a match in the bottle and place the hard-boiled egg on top and watch it get sucked in.

How To: Perform Vacuum Filtration in the Chemistry Lab
Find out how everything in a chemistry lab works, from pipettes to burners to recrystallization to storage. You'll get precise instructions on how to work and perform certain scientific duties in the chem lab, whether it's chemical or just ordinary high school science.

How To: View the universe on a Meade DS-2000 Series Telescope
All you science and astronomy nuts out there, pay attention, this detailed video tutorial series will tell you everything you need to know about using the Meade DS-2000 Series Telescope to ogle the universe.

How To: Perform the Barus effect
Watch this instructional science video to observe the Barus effect in action. A dyed solution of POLYOX (polyethylene oxide - it is the stuff used as the lubricant on the strip found in all modern razors) is extruded from a 50 mL syringe. On exit, a marked swelling in the liquid stream several times the diameter of the orifice is observed. The effect is referred to as the Barus Effect, but it also goes by several other names including the Merrington Effect, Die Swell, and Extrudate Swell.

How To: Draw blood from rats with two techniques
This video tutorial will show you the process of drawing blood from rats through the saphenous vein and by cardiac puncture.

How To: Make mothballs jump
If you put mothballs in a solution of vinegar and baking powder, the balls will collect air bubbles; then after they cause the balls to rise, the bubbles pop, and the mothballs drop.

How To: Do a demonstration of the Doppler effect
This video is an excellent example of how to demonstrate the doppler effect in the classroom.

How To: Karate Chop a Paint Stick with Help from Air Pressure and a Plastic Bag
How heavy is a plastic bag? Not very heavy at all, but in order to use a plastic bag to help me karate chop a thin wooden stick, I don't need weight—I need air pressure. Below, the "Quick Clip" demonstrates the power of air pressure via the vacuum created between a plastic bag and the countertop.

How To: Turn a Glass Bottle into an Electric Tornado Machine
We've all seen the classic tornado-in-a-bottle science experiment, which uses 2 two-liter bottles to create a whirlpool effect. This version requires you to get the tornado started yourself by spinning the bottles, but what if you want to make it fully automated?

How To: Make Your Own Glowing Green Fluorescein (Fluorescent Dye)
Fluorescent dye can be a great addition for decorating around the house for Halloween, especially for a haunted one. Creating your own fluorescent dye is a simple experiment, as long as you've got the proper chemicals and safety gear. Nurd Rage details the chemical process of creating your own fluorescein below.

How To: Make Trippy Triboluminescent Crystals That Glow Red or Blue When You Smash Them
If you're a Breaking Bad junkie who can't wait for the next episode, satisfy your craving with a little at-home chemistry and make some blue DIY smash-glow crystals! No, this is not Walter White's so-called "Big Sky" or even the subpar cringe-worthy product of his competitors. It's not even the same kind of crystals, otherwise you wouldn't be reading this. This is totally legal, even kid-friendly if you play it safe, though it actually requires more safety precautions than the potassium nitra...

News: This DIY Passion Fruit Battery Can Power Lasers!
It's been proven over and over that you can make batteries out of fruits and vegetables such as lemons, potatoes, and even apples. Turns out, passion fruit is also acidic enough to power a battery, but Maui Makers member Ryan K decided to take it a step further by adding a laser. Passion fruit, or Lilikoi as it's called in Hawaii, is usually over-abundant in Maui, so Ryan decided to put it to good use. Using anodized bolts, copper pipe, wire, switches, and some LEDs, he built a battery that c...
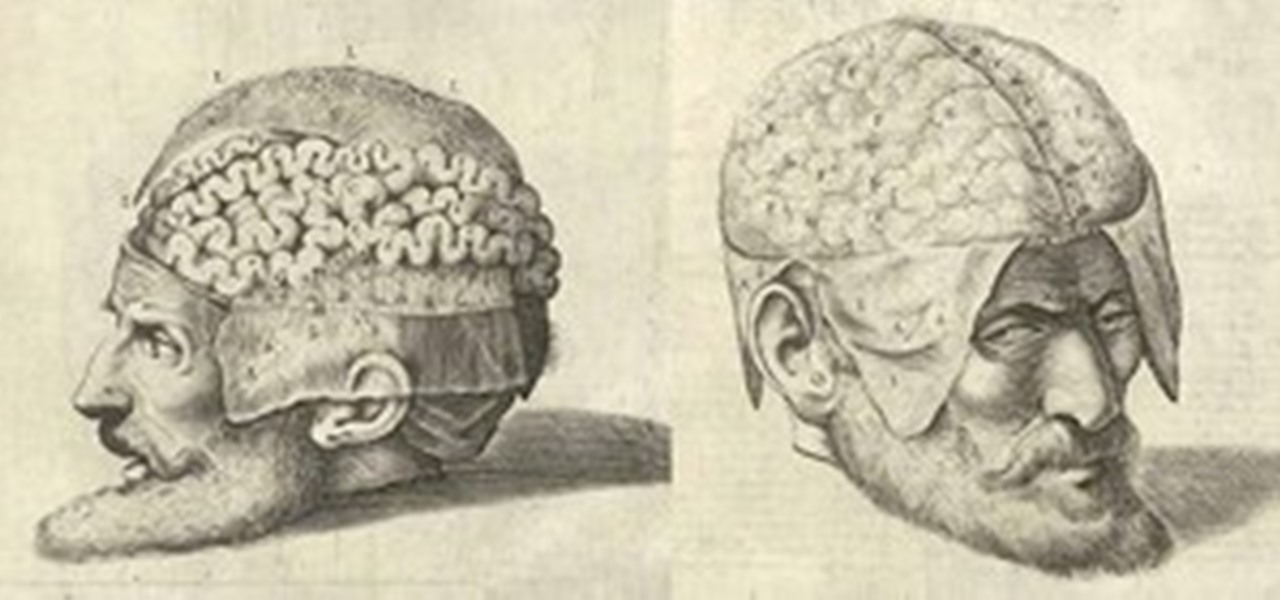
News: Dissecting a Human Head Through Anatomical Illustrations
Human anatomy is something every physician must undergo as a medical student. Some move on to become great doctors, some move on to become great artists, helping to better educate students and improve upon many illustrated representations of the human body since the days of medieval medicine. But thankfully, you don't have to be in the medical profession to enjoy the beautiful art of the human body created for teaching purposes.

DIY of The Decade: Pixar's UP House Comes to Life
Truly spectacular and one the most breathtaking DIY endeavors to grace the front page of WonderHowTo, a recreation of Pixar's UP house was launched to an altitude of 10,000 feet in a private airfield near Los Angeles this past week. The project was executed by a team of engineers, scientists and veteran balloon pilots—(meaning, please, Do Not Try This at Home).

How To: Observe the Full Moons in 2011 (Lunar Calendar)
When's the next full moon? Every time I've looked at the moon, I've wondered if it was full. Sometimes it looked like it was, but it's really hard to tell for sure unless you know the exact date of the full moon. Seriously, there's at a couple days before and after the full moon that could easily pass as full. At least, to someone like me who isn't naturally in tune with the moon's cycle (and doesn't have perfect vision).

How To: The Trick to Stabbing a Balloon Without Popping It
A balloon's worst fear is a sharp object, so usually when you puncture a balloon, it pops in your face. Not with this science trick! To puncture a fully blown up latex balloon without popping it, you'll need a pointed metal or wooden skewer and some plain old dish soap. That's it.

How To: Weigh something you can't see with this cool science trick
With this cool science trick by Mr. G, learn how to weigh something you can't even see! Using a stick, some string and two balloons, you can see in your very own home that air, invisible as it is, has weight. This is a good example to show atmospheric pressure.

How To: Make hydrazine sulfate with the hypochlorite and the Ketazine process
Hydrazine sulfate has many uses, but most notably, it's been used under the trade name of Sehydrin, a treatment for anorexia, cachexia and some even think cancer. But for we DIY chemists, it's useful for something entirely different— as a substitute for the more dangerous pure liquid hydrazine in chemical reactions. NurdRage shows you how to make it via some hypochlorite and the Ketazine process.
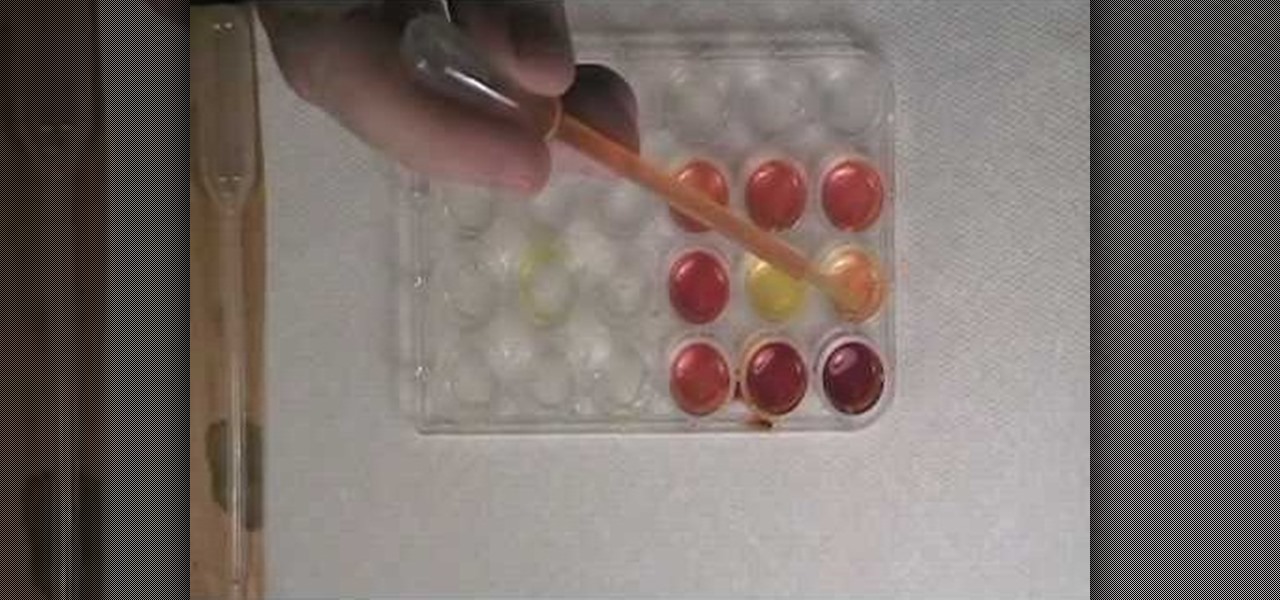
How To: Test for boron with curcumin
Want to test whether the soil sample or a water sample contains boron, watch this video. As shown in the video, the first step is to create test specimens of various concentrations using a simple turmeric and ethanol solution as a base for comparison. The basis of this experiment is the fact that curumin present in turmeric reacts with boron to form a reddish compound and so the result of this reaction can easily be verified visually.

How To: Make chlorine gas with pool chlorine and hydrochloric acid
Chlorine gas is a very useful oxidant, which was first introduced as a toxic weapon by the German Army. Even today, it's still used as a weapon, most recently in the Iraq War by insurgents. But chlorine gas has more useful (and less lethal) applications, and if you want to learn how to make some at home, NurdRage has the answers.

How To: Build parachutes with household materials
In this video, we learn how to build parachutes with household materials. You will need: a long string, Mylar, mesh, plastic, scissors, tape, and a weight. To get started, you will first cut your plastic or mesh into a circle or octagon shape. Then, take your string and cut it into several pieces. Then, tape the strings onto your material all around. After this, tie your weight to the strings so it's fully secured. Now, go to somewhere high and throw down your item! The parachute will help ke...

How To: Build a catapult out of rubber bands & a wire hanger
In this tutorial, we learn how to build a catapult out of rubber bands & a wire hanger. To do this, you will first need: one wire hanger, plastic spoon, rubber bands, small marshmallows, and measuring tape. Now, hold your hand in the center of the hanger and pull up both sides around it. After this, fold back the front for balance and straighten it up so it's stable. Next, you will take one rubber band and put it around the sides. Then, take the spoon and put it in between the rubber band and...

How To: Make ammonium chloride from hydrochloric acid and ammonia
This free video science lesson from the Home Scientist demonstrates a simple technique for creating ammonium chloride from hydrochloric acid and ammonia. For all of the relevant details and detailed, step-by-step instructions, as well as to get started trying this experiment yourself, watch this home-science how-to.

News: It's Hot, It's Creamy. It's Three Course Gum as a Meal
Remember Willy Wonka's magical gum? Wonka promised the flavors of tomato soup, roast beef, baked potato, blueberry pie and ice cream. As the avid gum lover Violet Beauregarde tested it out, she exclaimed: “It’s hot and creamy, I can actually feel? it running down my throat!” Um, yum... I think. Good news. Wonka's three course chewing gum is finally a reality-in-the-works. Scientists at the Institute of Food Research (IFR) have been developing recent advances in nanotechnology, which could pot...

How To: Properly balance chemical equations
In this video, we learn how to properly balance chemical equations. You will see what substances are in each side of the equation. You will then need to count the atoms that are on each side of the chemical equation. Once you write down the number of atoms on each side, you will need to even them out. To do this, you need to change the coefficients within the equation. These change the total number of atoms inside of a chemical. Once you do this, you will add more to each atom present and the...
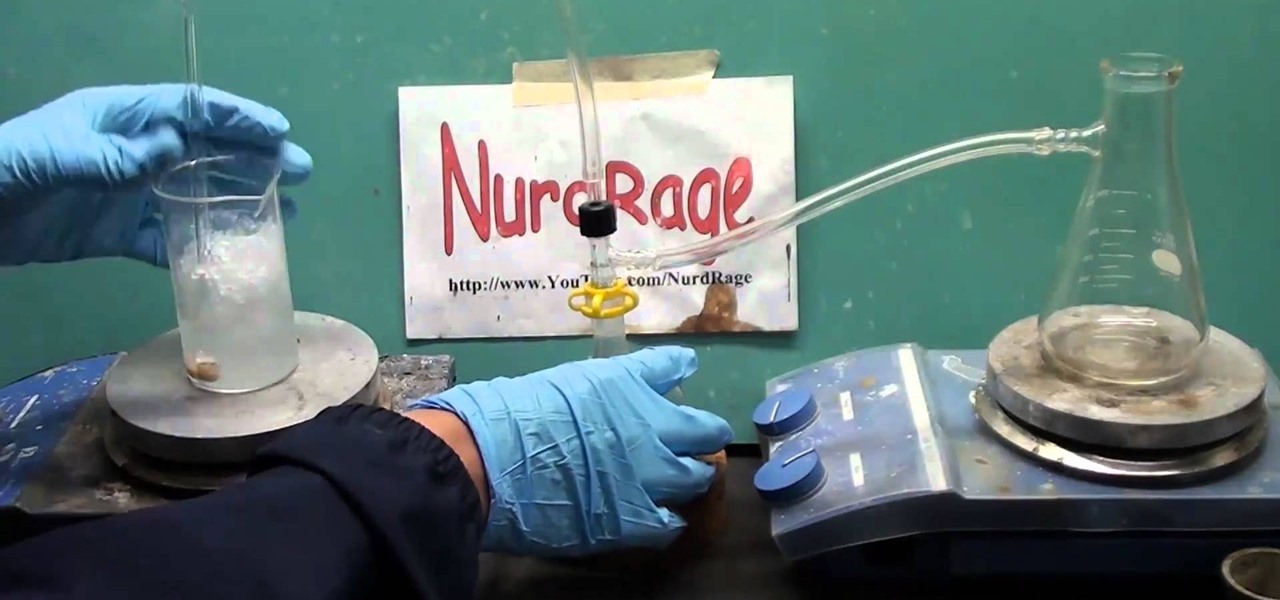
How To: Make hydrochloric acid from sodium bisulfate and table salt
In this home-science tutorial from WonderHowTo favorite Nurdrage, you'll learn how to create hydrochloric acid using 140 grams of sodium bisulfate, a pH lowering compound available in most pool stores, and 60 grams of sodium chloride salt and an external heat source. Watch for a full demonstration of the process and complete, step-by-step instructions.

How To: Synthesize copper(II) carbonate & sodium bicarbonate
This actions is a video tutorial in the Education category where you are going to learn how to synthesize copper(II) carbonate & sodium bicarbonate. For this you will need copper sulphate which is available in root kill and sodium bicarb which is baking soda. Take 100g of copper sulphate and dissolve in about 400ml of water. Now take 69.27g of baking soda. Add baking soda very slowly and keep stirring the solution. You got to be very careful as the chemical reaction will produce lot of carbon...

How To: Make elemental sulfur (sulphur)
This video tutorial is in the Education category which will show you how to make elemental sulfur (sulphur). The chemicals you need are nitric acid and sodium thiosulphate. The reaction produces toxic SO2 gas so keep it coved with a watch glass. Put 12.9 grams of sodium thiosulphate in a beaker and dissolve it in minimum amount of water. Pour about 15ml of nitric acid in to the beaker. Let it sit in a warm place for a couple of hours and the sulphur will settle at the bottom of the beaker. Th...

News: Jane Goodall Was a Babe (and One of History's Greatest Conservationists)
National Geographic recently published a retrospective of the lovely Jane Goodall, one of the world's most accomplished conservationists. The feature includes every image of Goodall to ever appear in the magazine for the past fifty years.

How To: Make green fire from household materials
In this video, we learn how to make green fire from household materials. You will need: antifreeze, boric acid, and a glass jar. First, pour the antifreeze into the jar until it's 1/3 of the way filled. After this, drop a spoonful of the boric acid into the mixture and mix it up thoroughly. Now, let it sit for 30 seconds and then go into a dark room. Light this up, then watch it burn! You can also put it into a spray bottle and spray it over a flame to see the green fire right in front of you!

News: Hand Fully Submerged in Liquid Nitrogen (OUCH... Right?)
Apparently submerging a hand in liquid nitrogen isn't as painful as one might suspect (if you trust the Leidenfrost effect as much as Theodore of Gray Matter):

How To: Build a cool soda bottle water rocket
Real rockets require top ingenuity from physics and engineering experts, but you can construct a smaller replica that costs almost nothing can can be made from materials you already have at home or can find at a hardware store.

How To: Turn pennies into silver and gold coins with zinc
One of the most famous and repeated chemistry experiments involves money. Some would say this is more of a trick than an experiment, but you can be the judge of that. No one can just turn pennies into silver or gold coins, but someone with a few chemicals can. So, if you want to cooler cents in your pocket, try out this chemistry trick yourself. Nurd Rage (Dr. Lithium) shows you how to turn pennies into silver and gold coins using zinc.

How To: Measure the volume of a balloon
Here we will demonstrate how to measure the volume of a balloon. A balloon is not a straight edged polygon shape, usually, so the mathematical equations get that much harder, on the flip side, it may be a spherical or ovalish shape, but measurements with math alone are detrimental due to the uneven sizes of the balloon. Here is how to do it properly. You will need a bucket, preferably, to hold water, a larger container than your original bucket, and a measuring container. Place the bucket ins...

How To: Find the North Star from the Northern Hemisphere
In this how to video, you will learn how to find the North Star in the northern hemisphere. To do this, you must know how to locate the Big Dipper and the Little Dipper. The Big Dipper is one of the brightest constellations in the sky. You can use this constellation to find the location of the north star. The two stars at the cup of the Big Dipper will point towards Polaris, also known as the North Star. The North Star is the end of the handle of the Little Dipper. This will be helpful if you...

How To: Determine the air amount in packing peanuts w/ acetone
How much air is actually inside a packing peanut? There is a simple experiment to conduct to see what is leftover. To begin, pour 200 ml of acetone into a glass container and add approximately 1800 ml of packing peanuts. Stir with a wooden spoon to dissolve. The video's author starts with the 1800 ml and realizes that that isn't enough. He then keeps adding until he has added an entire large bucket to the original 200 ml of acetone. After all are dissolved, all that is left in the glass conta...

How To: Make a Sugar Snake with Sulfuric Acid and Sugar
Sugar (a carbohydrate) is dehydrated with concentrated sulfuric acid. Since a carbohydrate was once considered just hydrated carbon, if you remove the water, carbon would be left over. The acid rips the water out of the sugar and the heat generated by this reaction causes the water to turn to steam. A black mass of carbon is produced.

How To: Make Ammonium Nitrate with NaHSO4 and Nitrate Salt
OK, so you don't necessarily have to make ammonium nitrate to have ammonium nitrate — ammonium nitrate can simply be obtained from fertilizers and instant cold packs. Making homemade NH4NO3 can be much more expensive then buying it, but this method is not meant to be a viable route, it's an exploration of science and chemistry.

How To: Reduce Static Electricity
Static electricity might seem like magic but you don't have to be a wizard to get rid of it. There are simple steps you can take to reduce the amount of static electricity in the air or on you by using some items you probably have at home.

How To: Make hot ice using Sodium Acetate Trihydrate
This video in the Education category will show you how to make hot ice using Sodium Acetate Trihydrate. For this purpose you will need a pan, 100g of Sodium Acetate Trihydrate, 25ml of water, a wooden spoon and a glass. Take the Sodium Acetate Trihydrate and put 100g in the pan. Then place the pan on a stove and turn to medium heat. The Sodium Acetate Trihydrate will start to melt. In about five minutes when it has melted fully, add 25ml of water. Let the solution simmer for two minutes while...