This video shows you how to draw the Lewis electron dot structure for the nitrite anion- first you start by drawing the symbols o, n and o, in that order, and then you proceed to draw two dots on the top of the n, and one on each other side.On the 'o' on the far left, you draw two dots on the top, and two dots on the left, putting just one dot on the bottom and the left- the right 'o' mirrors this pattern.Next you link all three letters with two lines, using the single dots on the left and right on all letters. You then draw another dot on the bottom of the right 'o', and put a negative in a circle diagonally down and right from the right 'o' then draw a line under the left 'o', connecting the dots on the 'n' and left 'o' to make a line. under this, write o=n-o, with two dots above the 'o' draw 4 dots on the left side of the left 'o', and 6 dots on all sides of the 'o' apart from the top left side. to the right of this, write a negative sign in a circle. you can also draw a mirrored version of this with an arrow pointing both ways (<->) then put these two mirroring pictures in brackets and your done~
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



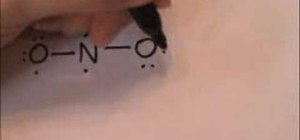



















Be the First to Comment
Share Your Thoughts