how to
Make Slime Without Borax: 5 Easy Recipes for Gooey Homemade Ooze


how to
How to Make Soap Out of Guava Leaf Extract for a Science Investigatory Project


how to
How to Make Your Own Homemade Glow Sticks



























Featured On WonderHowTo:
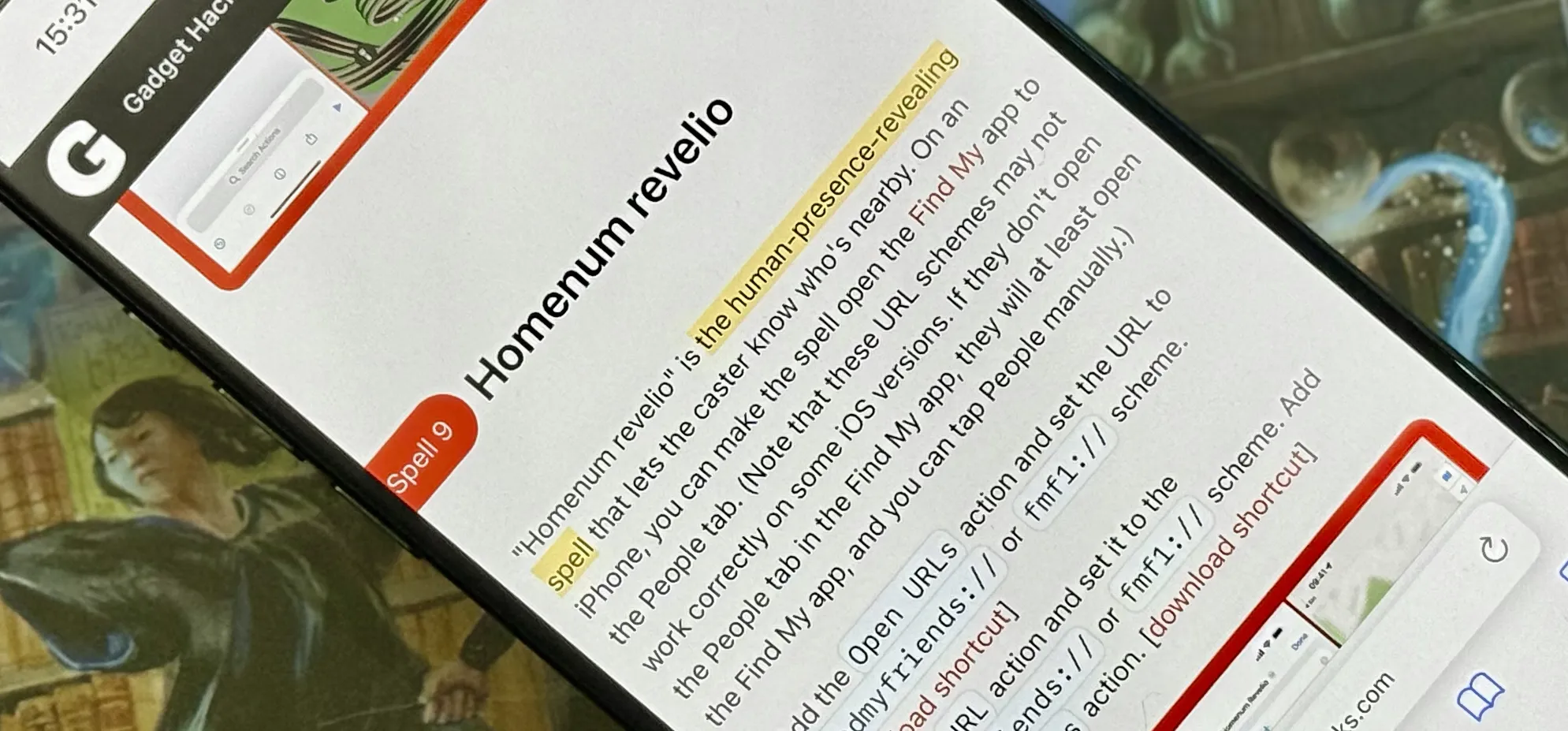
Productivity & Shortcuts










Featured On WonderHowTo:
Music & Audio










Featured On WonderHowTo:
Augmented Reality









Featured On WonderHowTo:
Gaming






















forum
RHNB-Milk 

forum
RHNB-Rice