A video which shows a reaction in which cobalt chloride reacts with water to form a hydrated form of cobalt and chloride ions. The reason for choosing this particular reaction for demonstrating chemical equilibrium is also explained. Cobalt chloride is dark blue in color. When water is gradually added to it, it changes color to pink as it undergoes the above mentioned chemical reaction. In the later part of the video, the reversible mature of this reaction is explained. When Calcium chloride is added to the mixture, the color again changes to dark blue as the hydrated cobalt again forms cobalt chloride solution.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



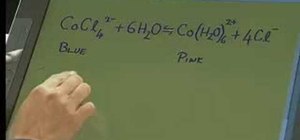



















Be the First to Comment
Share Your Thoughts