Not sure how to balance a chemical equation? According to the rules of chemistry, matter must be conserved in chemical reactions. When you balance a chemical equation, you account for all of the matter in the reaction.
What you will need:
• An unbalanced chemical equation
• Paper
• A pencil
Step 1: Count the number of atoms of each type
Determine how many atoms of each type are on each side of the equation.
Step 2: Find a starting element
Find an element that is in only one compound on the left side and in only one on the right side of the equation.
Step 3: Balance the equation for the first element
Multiply the chemical compound on the side that has less atoms of that type by the number required to achieve the same value on the other side.
Step 4: Continue balancing
Select another element or compound that is not balanced and balance it.
Step 5: Repeat as necessary
Repeat this process until the equation is completely balanced.
Step 6: Clear any fractions
Eliminate any fractions in the balanced equation by multiplying through by the denominator of the fraction.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.



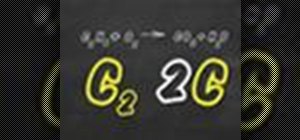




















Be the First to Comment
Share Your Thoughts